SCIENCE
CerFlux science advances human-relevant New Approach Methodologies (NAMs) that combine biomimetic engineering, microphysiological systems, and high-throughput ex vivo and in silico (computational and AI/ML) tumor models to better understand how individual tumors respond to therapy before treatment begins.
This page highlights peer-reviewed publications, high-impact conference abstracts, issued patents, and funded research that underpin platforms such as POET, BEST, SMART, and Lab-on-a-Brane technologies. Together, these efforts reflect a commitment to building human-relevant technologies that support more informed, patient-specific decision-making across translational cancer research and development.
Evaluation of a Novel Pan-RAS Inhibitor in 3D Bioprinted Tumor Models
De Nobrega, D; Eiler, LC; Ahirwar, P; Nallapu, S; Rawal, UP; Crawford, CL; Buchsbaum, DJ; Keeton, A; Maxuitenko, YY; Chen, X; Piazza, GA; Tsung, A; Budhwani, KI.
Publication | Cancers | 2025 | DOI: 10.3390/cancers17182958

Bioprinted 3D tumor models are an innovative approach that replicates the structure and environment of real tumors, offering an alternative to animal models for testing new drugs. In this study, we employ these models to evaluate a novel inhibitor targeting RAS proteins, common drivers of many cancers. By recreating the complex architecture of tumors in the laboratory, we demonstrate that this compound selectively eliminates tumor cells harboring RAS mutations while sparing cells without these mutations. Our work highlights the promise of 3D bioprinted tumor models for guiding drug development and advancing treatment strategies for cancers driven by RAS alterations.
Keywords: new approach methods; drug discovery; 3D bioprinting; colorectal cancer; RAS mutations; pan-RAS inhibitors; ex vivo tumor models
The Scienthetic Method: From Aristotle to AI and the Future of Medicine
Budhwani, KI.
Publication | British Journal of Cancer | 2024 | DOI: 10.1038/s41416-024-02841-1

While AI holds immense potential for accelerating advances in oncology, we must be intentional in developing and applying these technologies responsibly, equitably, and ethically. One path forward is for cancer care providers and researchers to be among the architects of AI and its adoption in medicine. Given the limitations of traditional top-down, hypothesis-driven design in an exponentially expanding data universe, on one hand, and the danger of spiraling into artificial ignorance (ai) from rushing into a purely ‘synthetic’ method on the other, this article proposes a ‘scienthetic’ method that synergizes AI with human wisdom. Tracing philosophical underpinnings of the scientific method from Socrates, Plato, and Aristotle to the present, it examines the critical juncture at which AI stands to either augment or undermine new knowledge. The scienthetic method seeks to harness the power and capabilities of AI responsibly, equitably, and ethically to transcend the limitations of both the traditional scientific method and purely synthetic methods, by intentionally weaving machine intelligence together with human wisdom.
Efficacy assessment of a novel pan-RAS inhibitor in KRAS-mutant and wild type colorectal 3D bioprinted organoid tumor tissue
Ahirwar, P; Charania, AA; Zuaiter, DR; Eiler, LC; Nizamuddin, A; Crawford, CL; Maxuitenko, YY; Piazza, GA; Budhwani, KI.
Abstract | Journal of Clinical Oncology | 2024 | DOI: 10.1200/JCO.2024.42.23_suppl.91
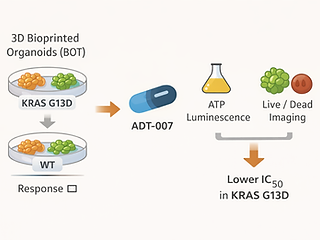
This ASCO meeting abstract evaluates ADT-007, a novel pan-RAS inhibitor, using a high-throughput ex vivo platform with 3D bioprinted organoid tumor (BOT) tissue models of colorectal cancer. The team generated BOTs from a KRAS G13D–mutant CRC cell line (HCT116) and a wild-type CRC line (HT29) and measured drug response via ATP luminescence plus high-content live/dead imaging. In these BOT models, ADT-007 showed a lower IC₅₀ in KRAS-mutant tissue than in wild-type tissue, consistent with separate in vitro/in vivo observations. The authors conclude that broadly acting pan-RAS inhibition, paired with BOT-based testing that better captures tumor microenvironment features, may help assess efficacy across RAS-driven cancers beyond a single KRAS allele.
Assessment of KRAS G12C inhibitors for colorectal cancer
Piazza, GA; Chandrasekaran, P; Maxuitenko, YY; Budhwani, KI.
Publication | Frontiers in Oncology | 2024 | DOI: 10.3389/fonc.2024.1412435

Colorectal cancer (CRC) is a common and often lethal cancer worldwide, and roughly 45% of CRC tumors carry activating KRAS mutations. KRAS is the most frequently mutated oncogene in humans (implicated in ~25% of all cancers), and these mutations can lock KRAS “on,” driving MAPK/AKT signaling that promotes uncontrolled growth, survival, and other features of malignancy. Although KRAS was long viewed as undruggable, the FDA has now approved two direct KRAS inhibitors—sotorasib and adagrasib—that covalently inactivate KRAS^G12C. These agents have shown meaningful benefit in KRAS^G12C-mutant NSCLC, but they’ve been far less effective in CRC, for reasons that remain incompletely understood. Because similar limitations may affect other mutant-specific KRAS inhibitors in development, it’s critical to understand the biologic basis of resistance. This review summarizes clinical trial results of KRAS^G12C inhibitors in CRC (as monotherapy and in combinations), outlines mechanisms that drive resistance, and discusses emerging RAS-targeting strategies designed to overcome or bypass these resistance pathways.
A comprehensive preanalytical protocol for fresh solid tumor biospecimens
Charania, AA; Pokal, AG; Zuaiter, DR; Crawford, CL; Esnakula, AK; Islam, M; Kim, AC; Budhwani, KI.
Publication | Methods | 2024 | DOI: 10.1016/j.ymeth.2024.06.005

Nearly seventy percent of diagnostic lab test errors occur due to variability in preanalytical factors. These are the parameters involved with all aspects of tissue processing, starting from the time tissue is collected from the patient in the operating room, until it is received and tested in the laboratory. While there are several protocols for transporting fixed tissue, organs, and liquid biopsies, such protocols are lacking for transport and handling of live solid tumor tissue specimens. There is a critical need to establish preanalytical protocols to reduce variability in biospecimen integrity and improve diagnostics for personalized medicine. Here, we provide a comprehensive protocol for the standard collection, handling, packaging, cold-chain logistics, and receipt of solid tumortissue biospecimens to preserve tissue viability.
Characterizing differential efficacy and phenotypic response to proteasome and survivin inhibitors in colorectal cancers using a high throughput organoid assay
Zuaiter, DR; Ahirwar, P; Pokal, AG; Patel, ZH; Charania, AA; Crawford, CL; Sewell-Loftin, MK; Tsung, A; Kim, A; Budhwani, KI.
Abstract | Journal of Clinical Oncology | 2024 | DOI: 10.1200/JCO.2024.42.3_suppl.153

This ASCO GI 2024 abstract describes a high-throughput, ex vivo “bioprinted organoid tumor” (BOT) platform designed to capture drug responses in 3D colorectal cancer tissue models beyond what standard monolayer and xenograft assays can predict. Using HT-29 colorectal adenocarcinoma cells embedded in a printable bioink, the team printed BOTs in different geometries and treated them with the proteasome inhibitor bortezomib and the survivin inhibitor YM-155, then assessed response via live/dead immunofluorescence and observed morphologic/phenotypic changes. Both agents produced a dose-dependent response, and importantly, the BOTs showed disrupted self-assembly/phenotypic modulation at and even below “effective” doses; signals that ATP-only assays can miss and that could lead to overstated efficacy when control wells proliferate differently. The authors conclude that functional, high-throughput ex vivo drug-response prediction platforms like BOTs could improve preclinical screening by capturing phenotype alongside viability.
SMART Microchamber Array for Multiplexed ex vivo Drug Screening
Budhwani, KI.; Budhwani, BK; Budhwani, KK.
Issued Patent | USPTO | US11833514B2 | Filed 2021 | Approved 2023

This issued patent strengthens the scientific and engineering foundation of Simple Microchamber Array Technology (SMART) by expanding the foundation for multiplexed, parallel exposure of intact biological samples to many test conditions in a single run. Building on SMART’s biomimetic microchamber array architecture, this patent further supports controlled, organized delivery of multiple fluids across defined regions of a single tissue specimen, enabling efficient side-by-side evaluation while preserving native tissue structure. The result is a practical path to higher-throughput, tissue-sparing workflows that can accelerate comparative compound assessment and iteration in human-relevant ex vivo testing, especially when patient material is limited.
Predictive efficacy biomarker for chemotherapy agents against triple-negative breast cancer bioprinted organoid tumors (BOTs) using solid tumor biopsy-on-a-chip
Bollenbecker, SE; Patel, ZH; Punjani, Z; Charania, AA; Patel, HK; Abott A; Kunkle, K; Sewell-Loftin, MK; Grossman, G; Budhwani, KI.
Abstract | Cancer Research | 2023 | DOI: 10.1158/1538-7445.SABCS22-P6-01-38

This SABCS 2022 / Cancer Research supplement abstract (P6-01-38) presents a way to expand triple-negative breast cancer (TNBC) tissue into 3D bioprinted organoid tumors (BOTs) that mimic core biopsies for ex vivo chemotherapy sensitivity/resistance testing. The team bioprinted MDA-MB-231 TNBC cells in an alginate-based bioink into biopsy-like geometries, cured/crosslinked the constructs, and then loaded them into a solid tumor “biopsy-on-a-chip” platform for drug exposure with live/dead immunofluorescence readouts. They report an optimized crosslinking/geometry workflow compatible with the chip and demonstrate diffusion of small molecules into the bioprinted tissue to substantial depth, supporting multi-agent testing. Overall, the work argues that BOT-based ex vivo platforms could provide more clinically relevant efficacy signals than conventional in vitro models and help guide personalized chemotherapy strategies.
Simultaneous rapid preclinical therapeutic evaluation in a novel ex vivo bioreactor
Bollenbecker, SE; Patel, ZH; Punjani, Z; Charania, AA; Patel, HK; Saleh, MN; Budhwani, KI.
Abstract | ESMO Open | 2022 | DOI: 10.1016/j.esmoop.2022.100729

This abstract argues that preclinical drug testing is often poor at predicting human outcomes because standard static cultures and many animal models don’t capture the human tumor microenvironment, contributing to high clinical failure rates. Here we present a microfluidic ex vivo tissue model built with engineered microporous membranes (soft lithography and electrospinning) and multicellular healthy and tumorous microtissues using pancreatic and colorectal cell lines. Permeability testing showed selective transport supporting barrier/transport function. When “tumorous” tissue was connected upstream of “healthy” vascular and muscle tissue, we observed migration and invasion, and identified invasive subpopulations with more aggressive proliferation than cells that stayed at the original tumor site. Overall, we propose human-relevant, ex vivo models as a path toward faster, more comprehensive pharmacologic testing and more predictive, personalized treatment guidance.
A hitchhiker’s guide to cancer models
Budhwani, KI; Patel, ZH; Guenter, RE; Charania, AA.
Publication | Trends in Biotechnology | 2022 | DOI: 10.1016/j.tibtech.2022.04.003

Cancer deals a devastating one-two punch – every year – by claiming 8 million lives worldwide while cratering $2.5 trillion in economic impact. Over 40% of patients wipe out their entire life savings within two years of diagnosis. This is unsustainable.
Next generation cancer supermodels built on CerFlux ChipMux™ - including POET®, PEER®, POETRY, and PROPHET - can deliver a one-two counterpunch by enhancing our understanding of mechanisms of cancer and by deploying this knowledge to combat cancer at every stage: from protective measures to early diagnosis, optimal personalized therapy, and precision post treatment surveillance.
Each category – in vitro, in vivo, ex vivo, in silico – undergirding cancer models is in a state of renaissance resulting in a faster pace of new knowledge across the bench to bedside continuum.
Evaluating anticancer agents on 3D bioprinted organoid tumors (BOT) to reduce cost and accelerate therapeutic discovery
Patel, ZH; Bollenbecker, SE; Charania, AA; Punjani, Z; Patel, HK; Sewell-Loftin, MK; Saleh, MN; Budhwani, KI.
Abstract | Journal of Clinical Oncology | 2022 | DOI: 10.1200/JCO.2022.40.16_suppl.e13500

Despite advances in therapeutics, cancer remains the second leading cause of death worldwide, and next-generation cancer models could help change that. However, obtaining enough tissue for ex vivo precision and personalized medicine can be challenging by tumor type and biopsy site. Current expansion approaches such as patient-derived xenografts (slow to establish) and patient-derived organoids (limited by size/scale), have practical constraints. Here we present 3D bioprinted organoid tumors (BOTs) that mimic core needle biopsy tissue, aiming to reduce time, cost, and barriers to evaluating novel therapeutics. We report producing and applying BOT core biopsy tissue for ex vivo testing, validating 3D microarchitecture with high-content fluorescence imaging and custom image analysis, measuring diffusion of stains and mock agents to 200 μm depth, and quantifying spatially distinct drug activity within intact BOT cores using advanced image-processing modules.
Preanalytical protocols for improving access to live tissue diagnostics in remote and low resource settings
Charania, AA; Bollenbecker, SE; Patel, HK; Punjani, Z; Patel, ZH; Papachristou, G; Contreras, CM; Tsung, A; Budhwani, KI.
Abstract | Journal of Clinical Oncology | 2022 | DOI: 10.1200/JCO.2022.40.16_suppl.e13501

Nearly 70% of diagnostic lab test errors stem from variability in preanalytical factors—everything from when tissue is removed from the patient to when it is tested in the lab. Because preanalytical protocols are often not standardized, specimen integrity can be compromised. Establishing clear protocols would help remote collection sites reduce variability in specimen viability and integrity, improving test rigor and reproducibility and supporting innovation in live-tissue diagnostics for personalized medicine. Here we present findings from a study on how cold-chain logistics affect solid tumor specimens, evaluating transit time, container and wet-ice packing, and transport media composition. Overall, standardized preanalytics can strengthen diagnostic reliability and enhance the predictive value of next-generation cancer models for translational research and bedside applications.
Evaluating efficacy on tumor biopsy tissue ex vivo before treatment for equitable cancer care
Patel, HK; Bollenbecker, SE; Punjani, Z; Charania, AA; Patel, ZH; Papachristou, G; Contreras, CM; Tsung, A; Budhwani, KI.
Abstract | Journal of Clinical Oncology | 2022 | DOI: 10.1200/JCO.2022.40.16_suppl.e13502

Nearly half the world will be diagnosed with cancer, and more than 1.7 million new cases are diagnosed each year in the U.S. Worse, systemic therapy is ineffective in ~70% of patients because the drugs don’t match the patient’s tumor, creating major physical, emotional, and financial burden. Because tumors are heterogeneous, the same treatment can produce very different responses across patients, making personalized approaches - based on testing efficacy directly on a patient’s own tumor tissue - fundamentally better than today’s trial-and-error care.
Here we present findings from our low-cost, ex vivo personalized solid tumor biopsy-on-a-chip platform designed to rapidly evaluate multiple therapeutics on intact core biopsy tissue before treatment. Core biopsies were generated from xenograft and human tumor tissue using 18- and 20-gauge spring-loaded biopsy systems. We observed and quantified differential activity of anticancer agents versus mock drugs using custom image-processing algorithms.
Democratizing cancer innovation by reducing cost and complexity of cancer models
Punjani, Z; Bollenbecker, SE; Patel, ZH; Charania, AA; Patel, HK; Papachristou, G; Contreras, CM; Tsung, A; Budhwani, KI.
Abstract | Journal of Clinical Oncology | 2022 | DOI: 10.1200/JCO.2022.40.16_suppl.e13503

Despite advances in high-throughput screening, combinatorial chemistry, databanks, and computational models, drug R&D remains expensive and slow—often taking over a decade. Pharma companies spend nearly $90B annually on preclinical research and trials, yet about 90% of drugs that look effective in pre-human studies fail in human trials. A key reason is that many preclinical efficacy methods don’t faithfully recapitulate in vivo microenvironments, driving failed trials and major time and cost burdens.
We developed a patented “Lab-on-a-Brane” (LOB) that better recreates in vivo tissue microenvironments, including barrier and transport functions, enabling organ–capillary interface models. We expanded the platform to support an air–liquid interface (e.g., lung microvasculature), then extended it to a “tumor-train” to model migration and invasion. Finally, we transformed the approach into a scalable, clinically relevant ex vivo Simple Microchamber Array Technology (SMART) that can concurrently assess multiple regimens directly on patient tissue.
Multimodal human-relevant cancer supermodel and methods of using the same
Budhwani, KI.
Issued Patent | USPTO | Filed 2022 | Approved 2025

This patent covers a human-relevant, ex vivo drug response screening and prediction method that uses viable patient-derived tumor tissue with intact extracellular matrix (ECM) to evaluate cancer therapeutic candidates outside the patient. The workflow emphasizes standardized collection and storage to preserve viability, preparation for analysis, and 3D microarchitecture and tumor microenvironment characterization of both ECM and cellular fractions to inform correlation and engineering of tissue proxies. Therapeutic candidates are then assessed by direct exposure on live or engineered tissue, enabling functional response evaluation in a setting designed to better reflect tumor biology than simplified models. Overall, the method supports a more rigorous, translational path for human-relevant NAMs based drug screening and personalized evaluation while retaining the complexity of intact tumor tissue.
Spatially Resolved Multiplexed Therapeutic Delivery in Intact BC Biopsies
Budhwani, KI (PI).
Grant | Breast Cancer Research Foundation of Alabama | 2021

With continued support from BCRFA, this project focuses on characterizing the controlled delivery of multiple isolated fluid streams to spatially distinct regions of intact breast cancer core biopsies within POET®. The work emphasizes rigorous validation of localized exposure, demonstrating that different agents can be applied to defined areas of the same biopsy while preserving native tissue architecture. By enabling spatially resolved, parallel testing within a single patient sample, the study strengthens the scientific basis for multiplexed efficacy assessment in ex vivo breast tumor tissue. This capability also supports more efficient use of limited biopsy material while generating richer, region-specific response data from each specimen.
SMART Biomimetic array device and methods of using same
Budhwani, KI.; Budhwani, BK; Budhwani, KK.
Issued Patent | USPTO | US11097274B2 | Filed 2019 | Approved 2021

Simple Microchamber Array Technology (SMART) is our patented biomimetic microchamber array device and set of methods for multiplexed exposure of intact biological samples to an array of fluids in parallel using microchannels and open-top microchambers. The architecture enables simultaneous evaluation of multiple compounds or conditions on a single intact tissue sample, supporting side-by-side comparison while preserving tissue microarchitecture. After exposure, samples can be characterized for response, viability, and related phenotypes to support comparative assessment across conditions. By compressing many test conditions into a single run, SMART is designed to increase throughput, conserve scarce patient material, and accelerate R&D iteration for human-relevant evaluation workflows.
Biomimetic Interface Device and Methods of Using the Same
Budhwani, KI.
Issued Patent | USPTO | US10969383B1 | Filed 2018 | Approved 2021

This issued CerFlux patent builds on our Lab-on-a-Brane innovation to deliver a configurable, human-relevant way to model barrier and transport behavior under realistic flow. The invention uses modular cassettes, such as an engineered scaffold positioned between fluidic chambers, integrated into a closed-loop recirculating system where flow and pressure can be precisely tuned to approximate in vivo-like dynamics. Because the cassettes can be scaled and reconfigured, including multi-cassette arrangements, the platform supports a range of R&D workflows where interfaces matter, from transport and PK-style studies to head-to-head compound evaluation in physiologically meaningful conditions. This architecture is designed to help teams generate more decision-ready data by bringing controllable physiology into an experimental format that is practical to deploy and iterate.
Attenuating Pandemic Supply Shocks with 3D Printed Emergency Use Equipment
Budhwani, KI; Pierce, AT; Arora, N.
Preprint | Research Square | 2021 | DOI: 10.21203/rs.3.rs-1229836/v1

In this preprint and accompanying NIH 3D Exchange repository, we address a real-world translational challenge exposed by COVID-19: severe supply-chain disruptions that created critical shortages in personal protective equipment (PPE), even at well-prepared medical centers. The work focused on developing a pragmatic strategy for emergency response manufacturing: instead of 3D-printing entire large volume PPE (often too slow), use additive manufacturing to rapidly produce small, low-cost adapters that repurpose existing components. As a case study, we describe a simple, inexpensive adaptation of elastomeric half-mask respirators for emergency clinical use in high-risk settings, highlighting how fast, locally deployable engineering solutions can help buffer healthcare systems against future supply shocks.
Development of Personalized Oncology Efficacy Test (POET) for Breast Cancer
Budhwani, KI (PI).
Grant | Breast Cancer Research Foundation of Alabama | 2020

Breast cancer is highly heterogeneous, yet systemic therapy is still too often selected using generalized clinical factors rather than patient-specific evidence, leaving many patients exposed to rounds of ineffective treatment and avoidable toxicity. Through our BCRFA-supported work, CerFlux is advancing its Personalized Oncology Efficacy Test (POET®)to help bridge this gap by testing a patient’s own tumor tissue ex vivo against multiple therapeutics in parallel, before treatment begins. The goal is straightforward: identify which options are most likely to work for an individual tumor - and which are unlikely to benefit - so care teams can move faster toward effective therapy while reducing the burden of failed treatment. By bringing actionable efficacy insight closer to the start of care, this approach is designed to improve decision-making, conserve precious time, and support more personalized treatment pathways.
Development of Personalized Ex Vivo Predictive Technology for Rapidly Matching Patient Tumors with Chemotherapy Regimens Before Treatment.
Budhwani, KI (PI); Tsung, A.
Grant | National Cancer Institute at National Institutes of Health (NIH/NCI) | 2020

Pancreatic ductal adenocarcinoma (PDAC) remains one of the deadliest cancers, and clinicians still lack predictive tools to determine which systemic therapy is most likely to benefit an individual patient, often leading to trial-and-error care and unnecessary toxicity. In this grant-supported effort, CerFlux is advancing its Personalized Oncology Efficacy Test (POET®) to help match patients to the right regimen before treatment begins. Building on a patented biomimetic in-vitro platform for pharmacologic transport and pancreatic tumor microtissue modeling, the project aims to identify both effective and ineffective options for each patient in advance of clinical decision-making. The work is strengthened through a commercial–academic collaboration with The Ohio State University’s James Comprehensive Cancer Center, including planned machine learning to generate a quantitative “POET Score” that helps rank therapies for each patient.
Evaluating Population Density as a Parameter for Optimizing COVID-19 Testing: Statistical Analysis
Budhwani KI; Budhwani H; Podbielski B.
Publication | JMIRx Med | 2021 | DOI: 10.2196/22195

This JMIRx Med short paper examines whether per-capita COVID-19 testing can mislead policy decisions by ignoring population density (how closely people live and interact). Using publicly reported data for all 67 Alabama counties, the authors compare tests-per-capita against cases and find only a weak relationship (reported correlation r=0.28), while new cases concentrate more heavily in denser areas even when those areas have relatively lower testing when viewed through a density lens. The paper argues that density-agnostic reporting can create a false sense of securityand recommends realigning testing allocation toward higher-density regions to better match transmission risk.
Measuring surface and interfacial tension in situ in Microdripping Mode for electrohydrodynamic applications
Budhwani, KI; Pekmezi, GM; Selim, MM.
Publication | Micromachines | 2020 | DOI: 10.3390/mi11070687

Surface tension is what lets small insects seem to “walk on water”—and in electrohydrodynamic atomization (EHDA), the same physics strongly shapes droplet formation. In this paper, we present an in situ method to estimate surface and interfacial tension (S/IFT) directly within EHDA workflows (eg, electrospinning/electrospraying), where conventional techniques (pendant drop, ring/plate) are often impractical to apply. Our approach uses signal-processing algorithms to extract droplet frequency/periodicity in EHDA microdripping mode and maps those measurements to computational fluid dynamics (CFD) solutions to infer S/IFT during operation. We validate the method against published ranges across multiple reference interfaces and show it captures expected trends such as reduced surface tension with increasing surfactant concentration, while explaining offsets relative to traditional methods. Overall, this provides a practical measurement workflow for tuning EHDA-based processes relevant to microphysiological systems and drug-delivery manufacturing.
Bridging the Gap in Training and Clinical Practice in Sub-Saharan Africa
Saleh, MN; Naik, G; Mwirigi, A; Shaikh,AJ; Sayani, S; Ghesani, M; Asaria, S; Sohani, AR; Sayed, S; Moloo, Z; Budhwani, KI; Talib, Z.
Publication | Current Breast Cancer Reports | 2019 | DOI: 10.1007/s12609-019-00322-6

In this paper, we describe a growing gap in cancer care between low- and middle-income countries (LMICs) and high-income countries (HICs): LMIC settings often lack basic preventive and diagnostic services for early cancer, while HICs have greater access to novel diagnostic and therapeutic modalities. We argue that narrowing this disparity will require innovative technology, knowledge sharing, and sustained public–private partnerships that can bridge geographic and resource constraints. A key emphasis is the value of onsite and online training programs to strengthen regional capacity and translate modern oncology practices into routine care. We also share case studies illustrating practical ways these collaboration and training models can help close the gap.
Lab-on-a-Brane: Biomimetic interface device and methods of using the same
Budhwani, KI.
Issued Patent | USPTO | US10114010B1 | Filed 2016 | Approved 2018

This issued patent describes a biomimetic apparatus configured to simulate physiological conditions by providing configurable barrier and transport interfaces within a closed-loop fluid-flow assembly. The modular cassette-based architecture enables controlled flow and transport across an engineered interface to better approximate in vivo dynamics where barrier function matters. Designed to be reconfigurable and scalable, the platform supports human-relevant experimental workflows such as transport studies and compound/therapeutic evaluation in systems that depend on realistic interface behavior.
Novel Biomimetic Microphysiological Systems for Tissue Regeneration and Disease Modeling
Budhwani, KI; Oliver, PG; Buchsbaum, DJ; Thomas, V.
Book Chapter | Advances in Experimental Medicine and Biology | 2018 | DOI: 10.1007/978-981-13-0947-2_6

In this publication, we introduce a new class of biomimetic microphysiological systems that addresses a fundamental shortcoming of conventional lab-on-chip platforms: the absence of tissue-relevant porous barriers that govern real biological transport and interface behavior. By integrating electrospun nanofibrous porous scaffolds directly into microfluidic systems, we developed a lab-on-a-brane (LOB) architecture that more faithfully reproduces key structural and functional features of native extracellular matrix. This platform enables more realistic investigation of molecular transport, air–liquid interfaces, and tumor progression, establishing a scalable and versatile foundation for advanced disease modeling and translational research. The work underscores the CerFlux approach to building physiology-first systems designed to push beyond incremental improvements and redefine what predictive models can achieve.
